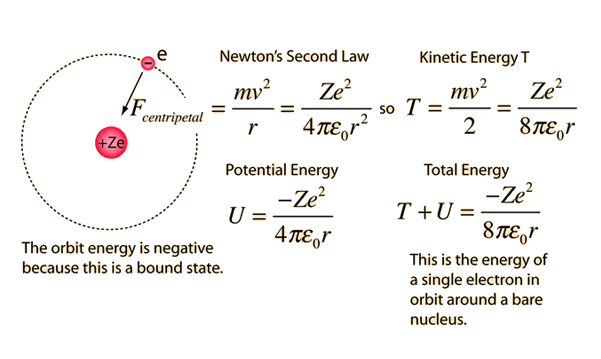
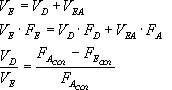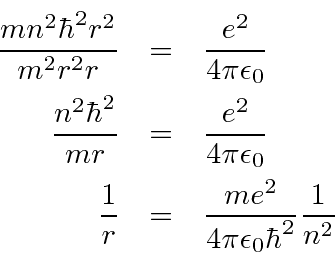Would the Bohr formula for the H-atom remain unchanged if proton had a charge (+4//3) e and electron a charge (-3//4) e, where e = 1.6 xx 10^(-19) C. Given reasons for you answer.
Using Bohr's formula for energy quantization, the ionisation potential of first excited state of hydrogen atom is: . (1) 13.6V, (2) 3.4V, (3) 2.6V, (4) 1.51V

Using Bohr's formula for energy quantization, the ionisation potential of the ground state of Li^++ atoms is?
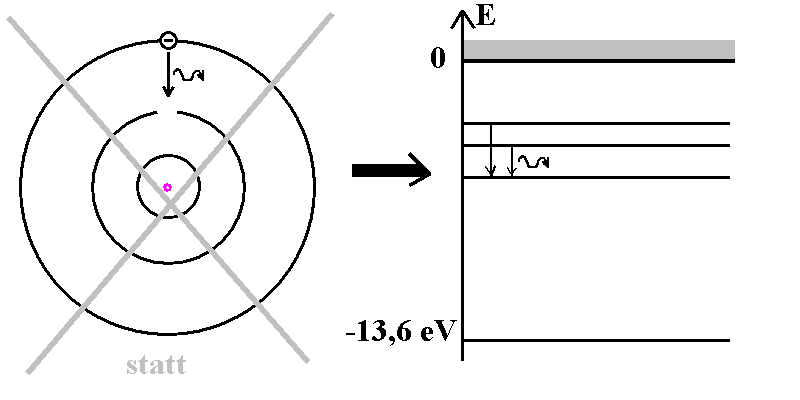

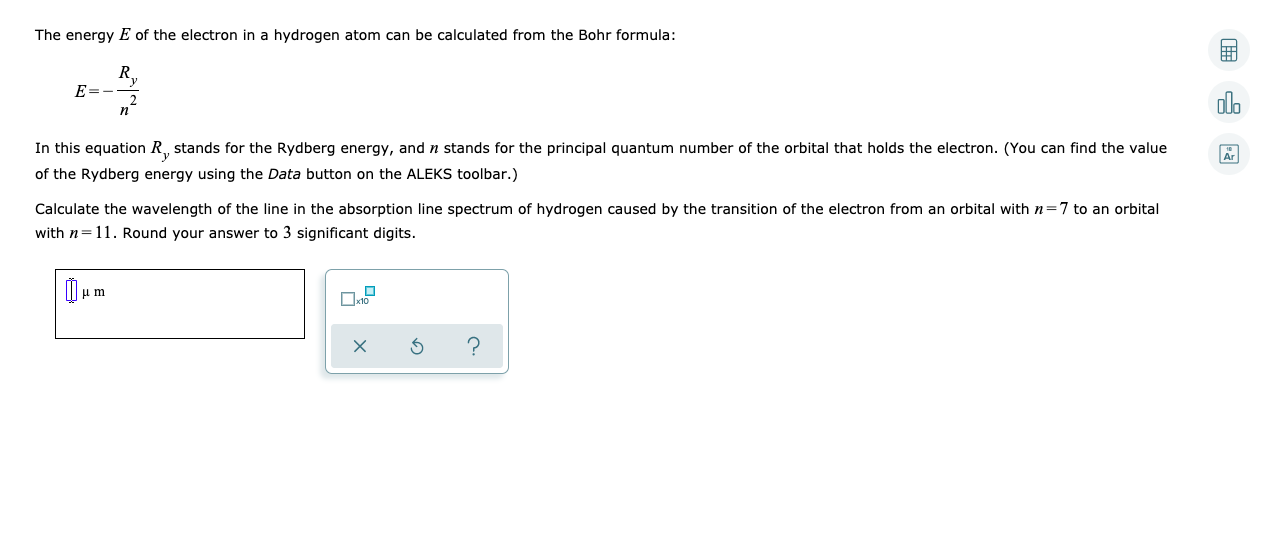
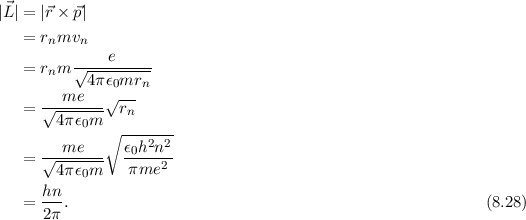



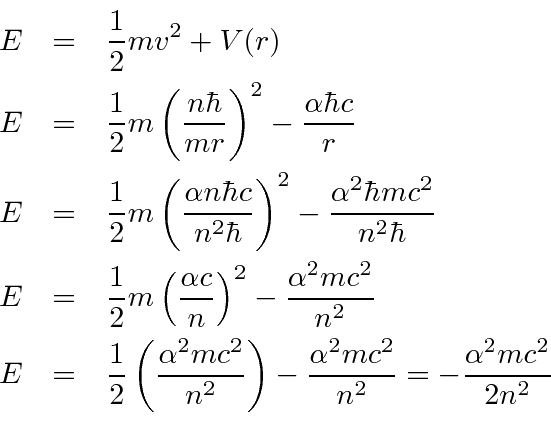




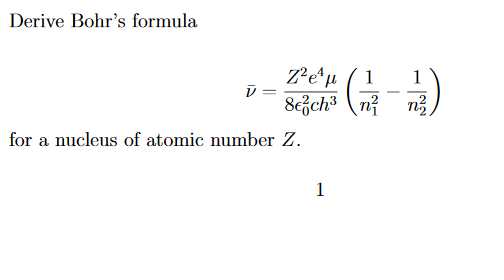



![Bohrsches Atommodell • Erklärung, Postulate, Formulierung · [mit Video] Bohrsches Atommodell • Erklärung, Postulate, Formulierung · [mit Video]](https://d1g9li960vagp7.cloudfront.net/wp-content/uploads/2020/03/Bohrsches-Atommodell_WP_3-2-1024x576.jpg)